What is C3H8O - Isopropyl alcohol?
Brand name: Auro-Dri
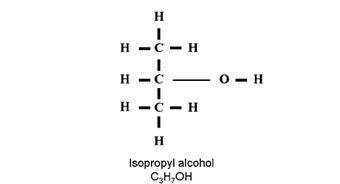
Formula: C3H8O
Density: 786.00 kg/m³
Boiling point: 180.5°F (82.5°C)
IUPAC ID: isopropyl alcohol
Molar mass: 60.1 g/mol
The Basics
Isopropyl alcohol is a colorless, combustible liquid; it is classified as a secondary alcohol, meaning that the carbon atom that makes up the alcohol component is bound to two other carbon atoms. C3H8O is made from combining water with propylene, and can be easily separated from its water constituent though a process called salting out. This involves the addition of an organic salt such as sodium chloride. [1]
Medicinal and Other Uses
In addition to providing an outlet for chemical experimentation in the lab, isopropyl alcohol can be found in nearly any home medicine cabinet. In fact, it was once the popular choice for medicinal rubdowns—hence the nickname "rubbing alcohol." When applied to the skin, the water component quickly evaporates, cooling the skin's surface and rapidly reducing body temperature. For this reason, it was once recommended for reducing fever in children. However, since rapid decrease in temperature is now considered detrimental, health care professionals no longer recommend the use of it for this purpose. [1]
Formula: C3H8O
Density: 786.00 kg/m³
Boiling point: 180.5°F (82.5°C)
IUPAC ID: isopropyl alcohol
Molar mass: 60.1 g/mol
The Basics
Isopropyl alcohol is a colorless, combustible liquid; it is classified as a secondary alcohol, meaning that the carbon atom that makes up the alcohol component is bound to two other carbon atoms. C3H8O is made from combining water with propylene, and can be easily separated from its water constituent though a process called salting out. This involves the addition of an organic salt such as sodium chloride. [1]
Medicinal and Other Uses
In addition to providing an outlet for chemical experimentation in the lab, isopropyl alcohol can be found in nearly any home medicine cabinet. In fact, it was once the popular choice for medicinal rubdowns—hence the nickname "rubbing alcohol." When applied to the skin, the water component quickly evaporates, cooling the skin's surface and rapidly reducing body temperature. For this reason, it was once recommended for reducing fever in children. However, since rapid decrease in temperature is now considered detrimental, health care professionals no longer recommend the use of it for this purpose. [1]
